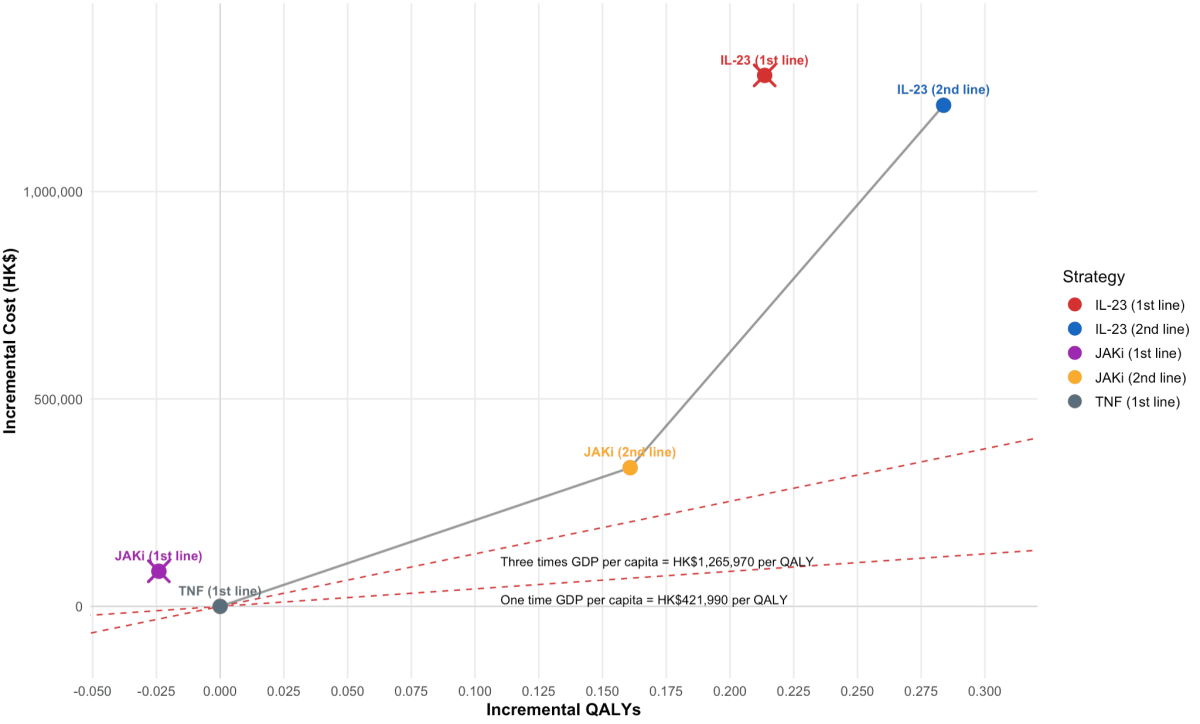
The cost-effectiveness frontier comprises TNF (1st line), JAKi (2nd line), and IL-23 (2nd line).
However, JAKi (2nd line) and IL-23 (2nd line), all exceed the 3× GDP per capita threshold.
Note: Abbreviations: QALY, quality-adjusted life-year. Interleukin-23 (IL-23) inhibitors: risankizumab, mirikizumab, guselkumab. JAK inhibitors (JAKi): upadacitinib. TNF, Tumor Necrosis Factor; GDP, Gross Domestic Product.
Each data point represents a treatment strategy for moderate-to-severe Crohn's disease compared with the reference (TNF inhibitors 1st-line → non-TNF biologics → best supportive care). The solid grey line is the cost-effectiveness frontier of non-dominated strategies.
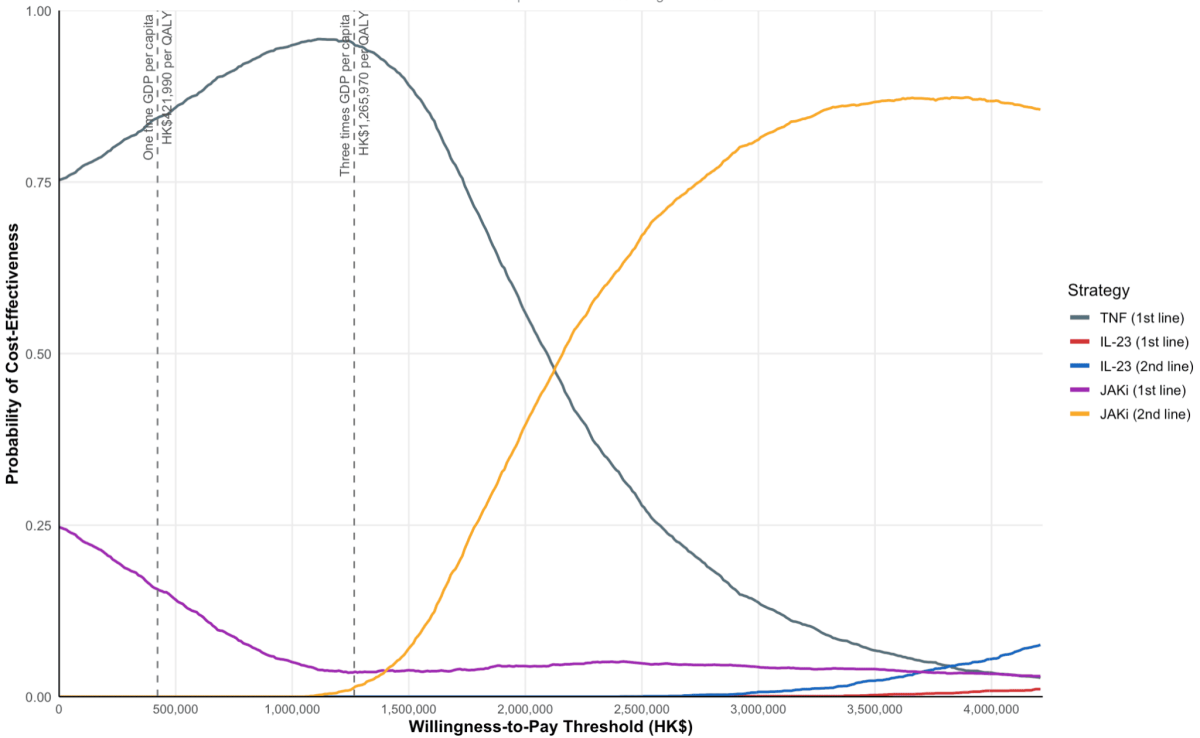
The CEACs summarise this uncertainty across different cost-effectiveness thresholds.
At a cost-effectiveness threshold of 3× GDP per capita, TNF (1st line), JAKi (1st line), and JAKi (2nd line) had a 95.1%, 3.6%, and 1.3% probability of being the most cost-effective option, respectively, while IL-23 strategies had negligible probabilities.
At a higher threshold of 10× GDP per capita, JAKi (2nd line) became the most cost-effective option.
Abbreviations: QALY, quality-adjusted life-year. Interleukin-23 (IL-23) inhibitors: risankizumab, mirikizumab, guselkumab. JAK inhibitors (JAKi): upadacitinib. TNF, Tumor Necrosis Factor; GDP, Gross Domestic Product.
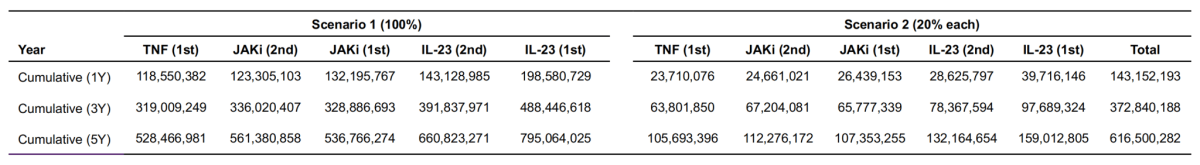
Two distinct market share scenarios were assessed to illustrate a range of possible uptake for novel among newly entering patients in the moderate-to-severe Crohn‘s disease (CD) treatment pathway:
Advanced therapy, including biologics and small molecules, and steroid dependency were used to identify patients with moderate-to-severe CD in CDARS. 463 patients with moderate-to-severe CD who had not received advanced therapy, undergone CD-related surgery, or died by end of 2024. Predicted incident cases with moderate-to-severe CD in Hong Kong during 2025 and 2029 are 63 per year. These patients were analyzed in the budget impact (Table 3).
Abbreviations: QALY, quality-adjusted life-year. Interleukin-23 (IL-23) inhibitors: risankizumab, mirikizumab, guselkumab. JAK inhibitors (JAKi): upadacitinib. TNF, Tumor Necrosis Factor; GDP, Gross Domestic Product.
