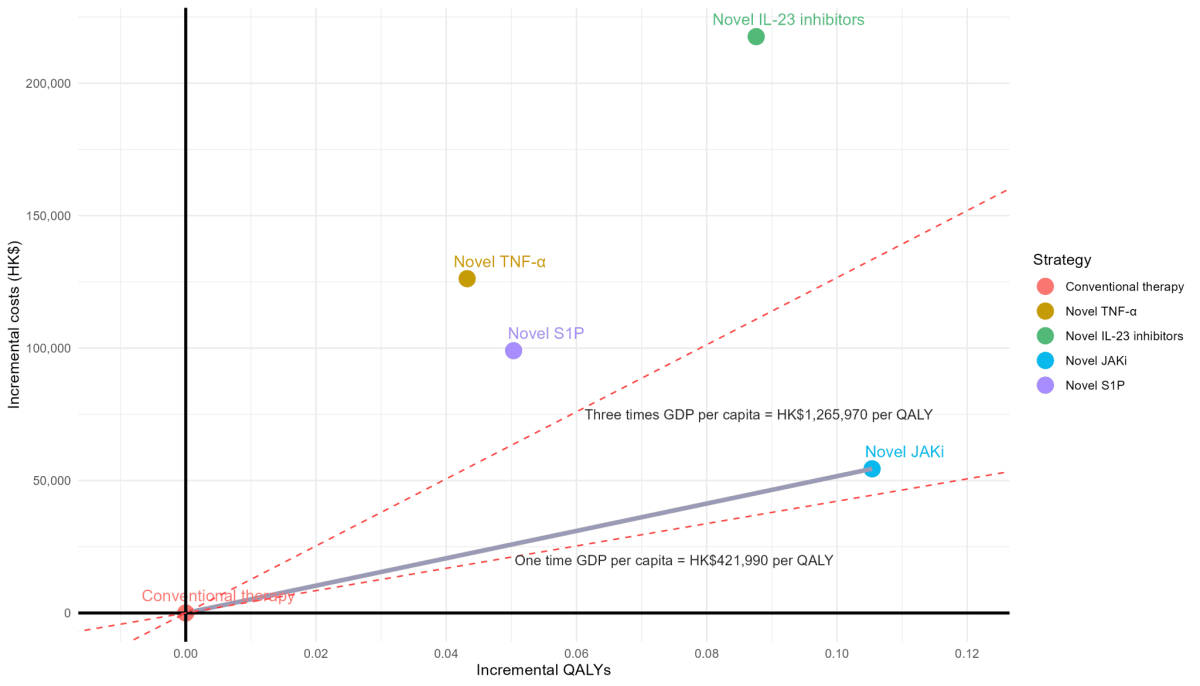
The cost-effectiveness frontier linked conventional therapy and novel JAKi (ICER: HK$516,472 per QALY) as the only dominant strategies, excluding other interventions due to inferior cost-effectiveness. Novel JAKi became cost-effective only when thresholds exceeded HK$516,472 per QALY, otherwise conventional therapy remained optimal.
Notes: Novel TNF-α (Tumor Necrosis Factor-α inhibitors) golimumab; Novel IL-23 inhibitors (Interleukin-23 inhibitors): mirikizumab, risankizumab, gueslkumab; Novel JAKi (Janus Kinase inhibitors): filgoticib, upadaticinib; Novel S1P (Sphingosine-1-phosphate receptor modulators): etrasimod, ozanimod.
QALY, Quality-Adjusted Life Year; GDP, Gross Domestic Product.
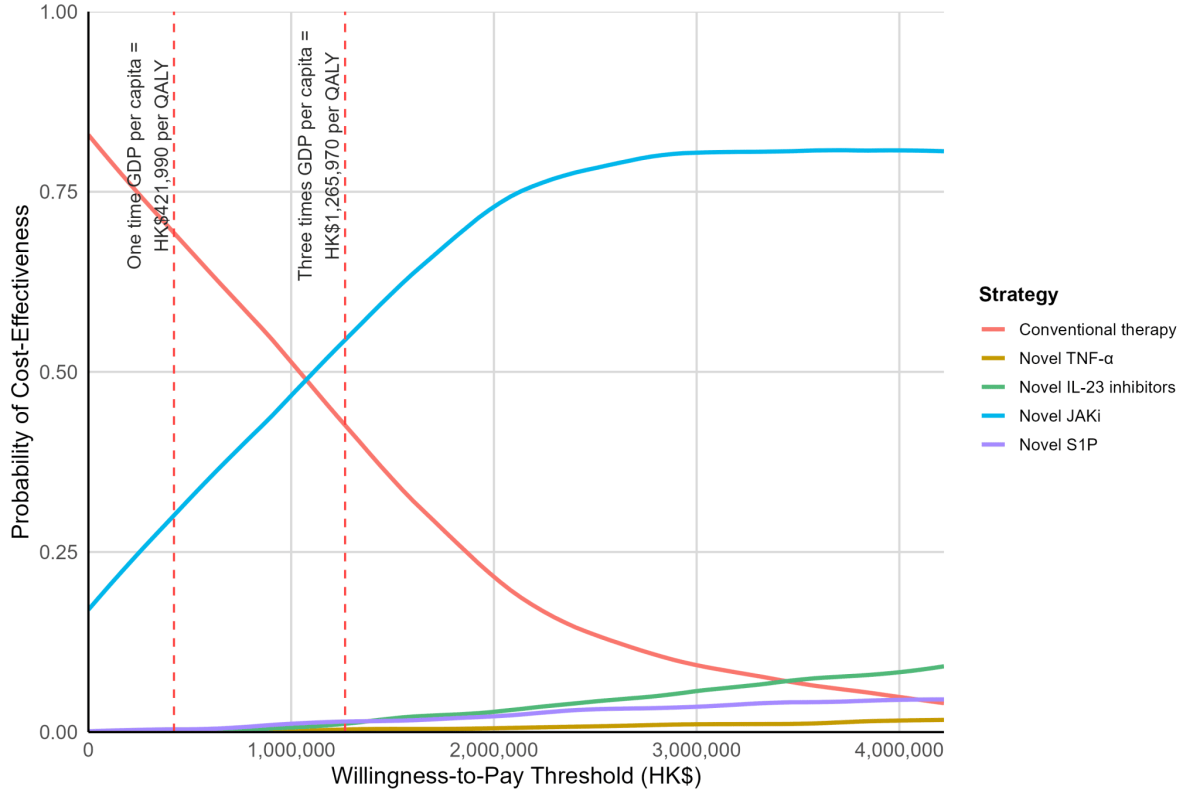
The CEACs summarise this uncertainty across different cost-effectiveness thresholds.
At cost-effectiveness threshold as three-time GDP per capita in Hong Kong in 2024, novel JAKi, novel IL inhibitors, and novel TNF-α has roughly a 54.4%, 0.4%, 0.1% and 0.1% probability of being the most cost-effective option.
At ten-time GDP per capita (cost-effectiveness threshold value = HK$4,219,900 per QALY), the novel JAKi becomes the most cost-effective option.
Notes: Novel TNF-α (Tumor Necrosis Factor-α inhibitors) golimumab; Novel IL-23 inhibitors (Interleukin-23 inhibitors): mirikizumab, risankizumab, gueslkumab; Novel JAKi (Janus Kinase inhibitors): filgoticib, upadaticinib; Novel S1P (Sphingosine-1-phosphate receptor modulators): etrasimod, ozanimod. QALY, Quality-Adjusted Life Year; GDP, Gross Domestic Product.
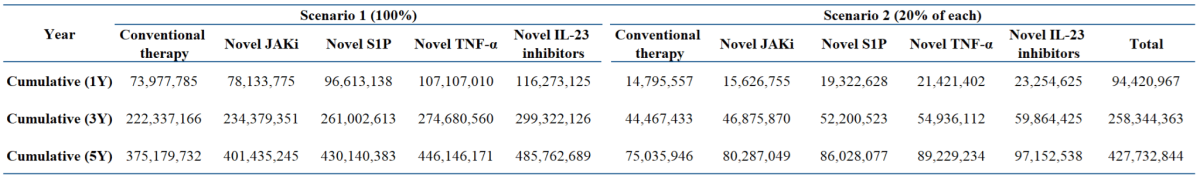
Two distinct market share scenarios were assessed to illustrate a range of possible uptake for novel among newly entering patients in the moderate-to-severe ulcerative colitis treatment pathway:
Advanced therapy, including biologics and small molecules, and steroid dependency were used to identify patients with moderate-to-severe ulcerative colitis in CDARS. 336 patients with moderate-to-severe ulcerative colitis did not receive advanced therapy within 180 days in Hong Kong in 2024. Predicted incident cases with moderate-to-severe UC in Hong Kong during 2025 and 2029 are 50, 55, 58, 60, and 61, respectively. These patients were analyzed in the budget impact (Table 3).
